Considerations for a revised adult cochlear implant candidacy evaluation protocol
*A. Biever, *^C. Amurao, ^M. Mears
*Rocky Mountain Ear Center, 601 E. Hampden Ave #430, Englewood, CO 80113, United States of America, ^Cochlear Americas, 10350 Park Meadows, Lone Tree, CO 80124, United States of America
Purpose
To compare two commonly utilized tests in cochlear implant candidacy evaluations, the Consonant- Nucleus-Consonant (CNC) test in quiet and the AzBio Sentences test in noise, for uniformity and appropriateness.
To recommend an updated streamlined test battery that relies on a monosyllabic word test and a self-report subjective measure of hearing capabilities.
Key messages
The CNC word test is efficient and effective in determining cochlear implant candidacy and can be used to monitor hearing performance over time.
The Speech, Spatial and Qualities of Hearing Scale (SSQ) or SSQ-12 furnishes subjective, real-world insight into an individual’s hearing difficulties and offers complementary information to objective speech understanding measures.
Methods
- Prospective, non-randomized, multicenter controlled trial at 13 clinical sites
- 100 experienced hearing aid users > 18 years old with postlinguistic onset of bilateral, moderate sloping to profound sensorineural hearing loss
- Aided CNC word score < 40% in the ear to be implanted and < 50% in the contralateral ear
Preoperative speech recognition was evaluated in the sound field for unilateral (HA) and bilateral (2HA) conditions and at 6-months postoperatively for unilateral (CI) and bimodal (CI+HA) conditions. CNC words in quiet at 60 dBA and AzBio sentences at 65 dBA in background noise at +10 and +5 dB SNR (speech and noise at 0° azimuth) were presented. In addition, participants completed the subjective Speech, Spatial and Qualities of Hearing Scale (SSQ), pre-(2HA) and 6-months postoperatively (CI+HA). All assessment results are based on matched pair data.
Results
In total, 100 subjects were implanted in this study, ranging from 23 to 91 years of age with a mean age of 67 years at the time of implantation. The duration of hearing loss in the ear to be implanted ranged from 1 to 66 years with an average duration of 27 years, and all subjects were experienced hearing aid users.
Of the 100 subjects who were enrolled in the study, 96 completed the speech testing at the 6-month endpoint. In the implanted ear, subjects demonstrated an average improvement of 46 percentage points on CNC words at 6 months postoperatively, and 28 percentage points and 11 percentage points for AzBio sentences at +10 dB SNR and +5 dB SNR respectively. The improvement on each of the three tests was statistically significant at the 6-month endpoint, and Figure 1 shows the distribution of individual scores. All results represent matched pair data.
More variation in individual outcomes for speech recognition scores for sentences in noise was observed compared to CNC words in quiet in both unilateral and bimodal listening conditions at each test interval. Competing background noise presents the potential for additional cognitive load for hearing-impaired individuals. The scores obtained on a complex sentence test in noise can sometimes reflect the central processing function of the patient, more so than capture their peripheral listening ability. Administration of a monosyllabic word test in quiet can more reasonably assure that the clinician is evaluating peripheral performance, without the contribution of additional central processes.
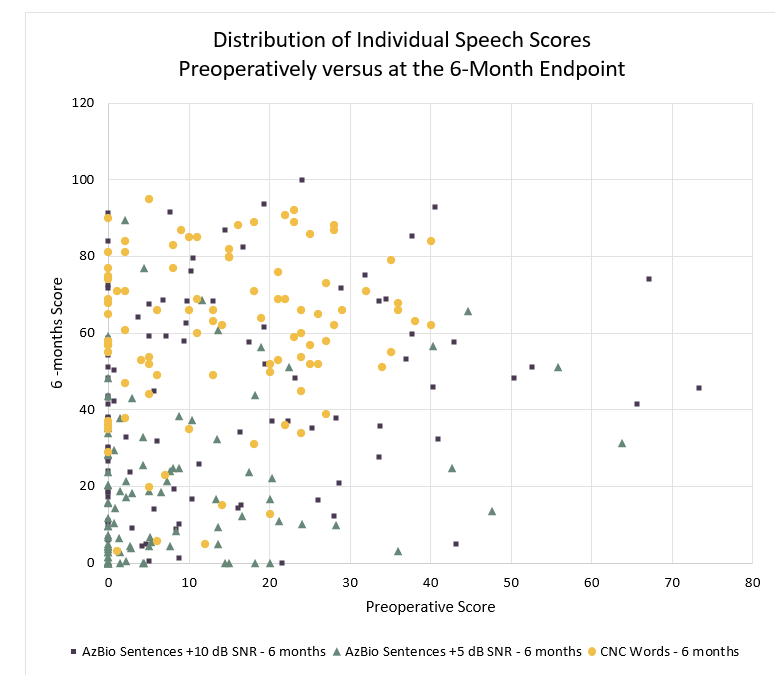
Figure 1: The distribution of individual speech scores preoperatively versus at the at the 6-month endpoint. Group mean score for CNC words significantly increased by 46 percentage points ( p<0.001). Group mean score for AzBio sentences scores at +10 dB SNR significantly increased by 28 percentage points (p<0.001). Group mean score AzBio sentences scores at +5 dB SNR significantly increased by 11 percentage points (p<0.001). Note the increased variability in scores on the AzBio sentences, as well as the floor effect seen most prominently in the +5 dB SNR condition. CNC indicates Consonant-Nucleus-Consonant; SNR, signal-to-noise ratio.
As shown in Figure 2, the overall total score on the SSQ at the 6-month endpoint (N=95) showed a clinically significant improvement from 3.2 to 5.7 (a 2.5 point increase) with 77% of subjects reporting an overall improvement. Results from the SSQ scenarios can be classified into pragmatic subscales that reflect self-reported difficulties associated with acoustically dynamic listening situations where binaural hearing is especially important. The mean pre-to-post speech in noise subscale change was 2.6 and the mean pre-to-post speech in quiet subscale change was 2.9 with 86% of subjects showing a significant increase.
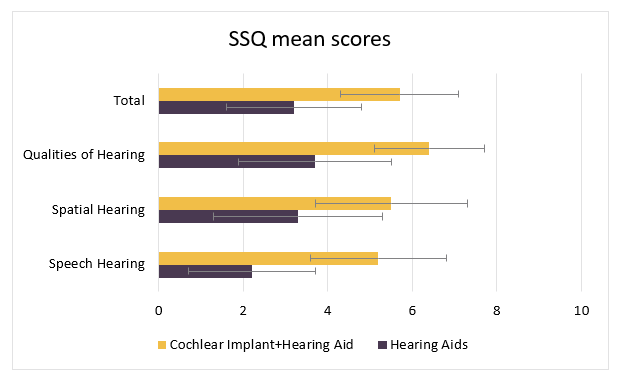
Figure 2: Mean scores on the SSQ (49 questions) preoperatively with hearing aids and at 6-months postoperatively with a cochlear implant & hearing aid. Significant postoperative changes were demonstrated in each subscale as well as the total score (p<0.001).
Conclusions
Results from this multi-center study suggest that clinicians adopt CNC words as the primary candidacy evaluation instrument rather than AzBio Sentences in noise. A subjective tool, such as the SSQ-12 or SSQ, can provide important complementary information regarding how the individual feels about everyday hearing abilities.
To read more Science Spotlights visit Cochlear ProNews.
Citation: Biever A, Amurao C, Mears M. Considerations for a revised adult cochlear implant candidacy evaluation protocol, Otol Neurotol. 2021 Jan; 42; (1),159-164 doi: 10.1097/MAO.0000000000002966
This article is intended to serve as a resource for clinicians to help keep up to date with current clinical literature and is intended for professionals only. Clinical literature is based on research, which may include the experimental use of new or currently available products and technologies. Therefore, literature presented on this blog may represent use of Cochlear products that does not align with the intended use or indications approved by regulatory bodies, also known as off-label use. Cochlear does not condone any off-label use of its products, and it is not Cochlear’s intent to promote off-label use by providing this blog as a resource to clinicians.
This article summary is for education and information sharing amongst professionals and partners. Any representations made in relation to medical devices or therapeutic goods are not advertisements. The representations and views expressed in this summary are those of the authors and do not reflect the views of Cochlear. Cochlear does not endorse any particular treatment or research protocol nor off-label use.”


